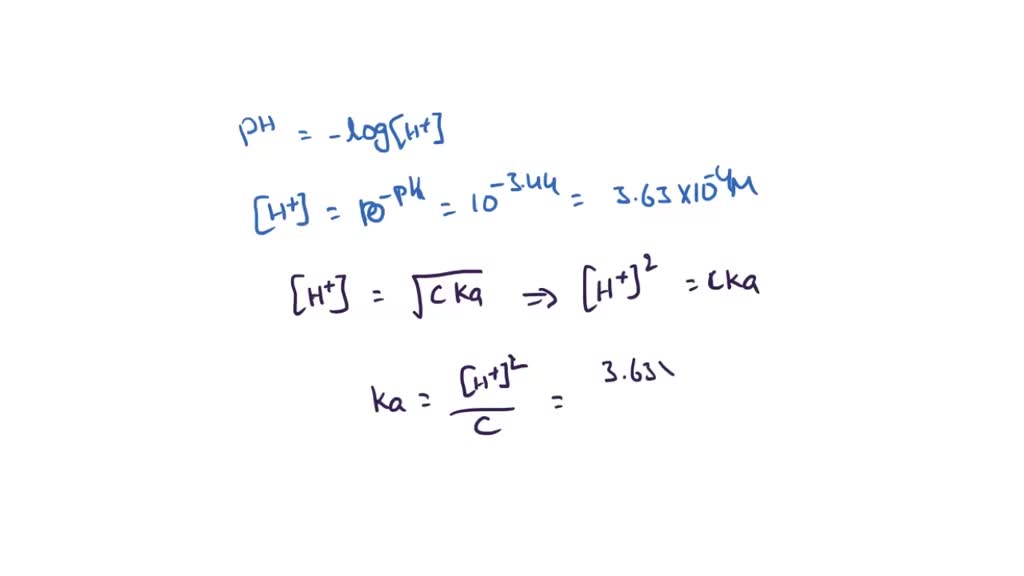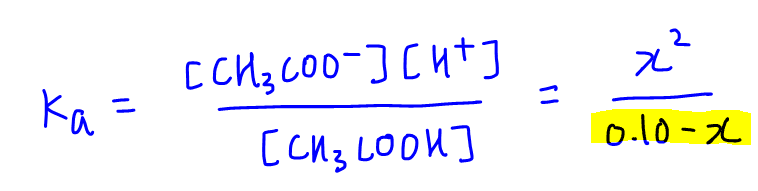SOLVED: Suppose the pH measure of how acidic or basic a water is) value of a soil in a certain area has the following distribution function: k(z2 20z + 100) , 3 <

Calculate the `pH` at which an acid indicator with `K_(a) = 1.0 xx 10^(-5)` changes colour when ... - YouTube

Calculate the pH of the solution formed when 45.0 mL of 0.100 M NaOH is added to 50.0 mL of 0.100 M CH3COOH (Ka = 1.8x10-5). | Homework.Study.com

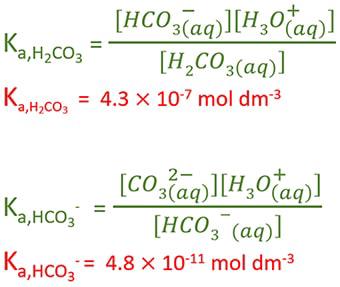
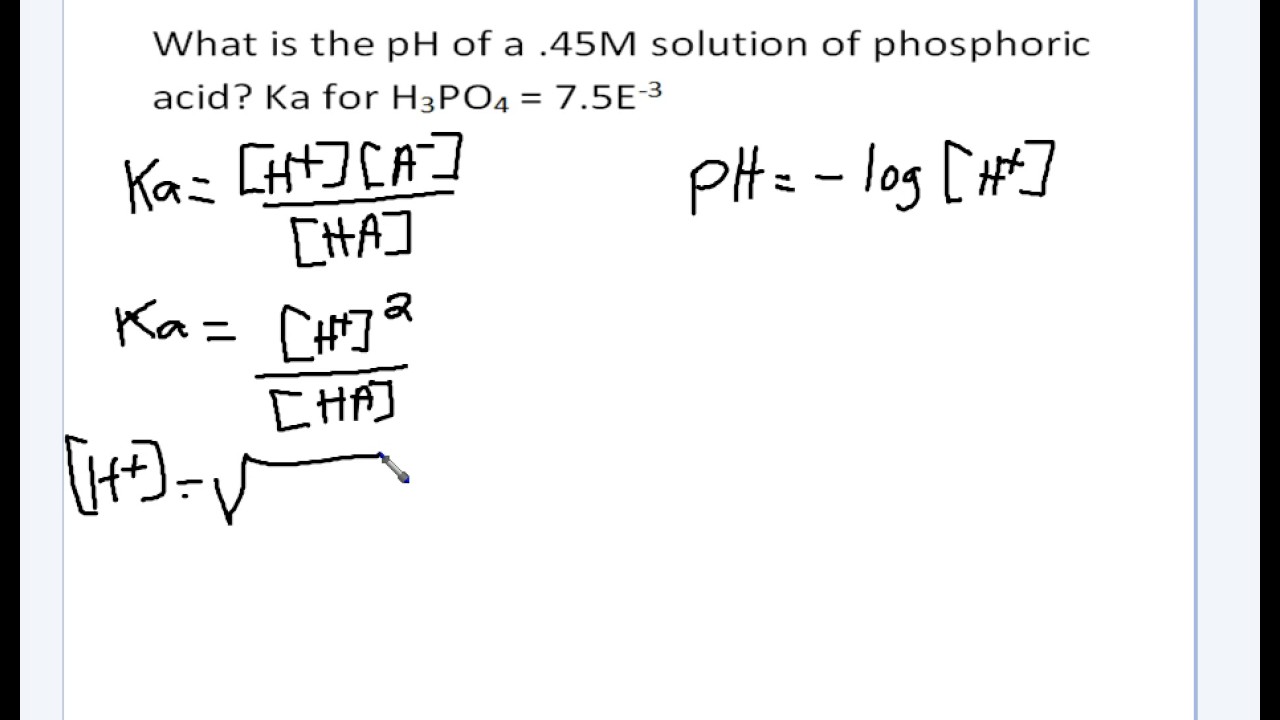
SOLVED: The pH of a solution depends upon the concentration of the acid (or base) as well as the extent of the reaction (2) as given by the equilibrium constant K: The

The Ka value for acetic acid, CH3COOH(aq), is 1.8x10^-5. Calculate the ph of a 2.80 M acetic acid solution - Home Work Help - Learn CBSE Forum
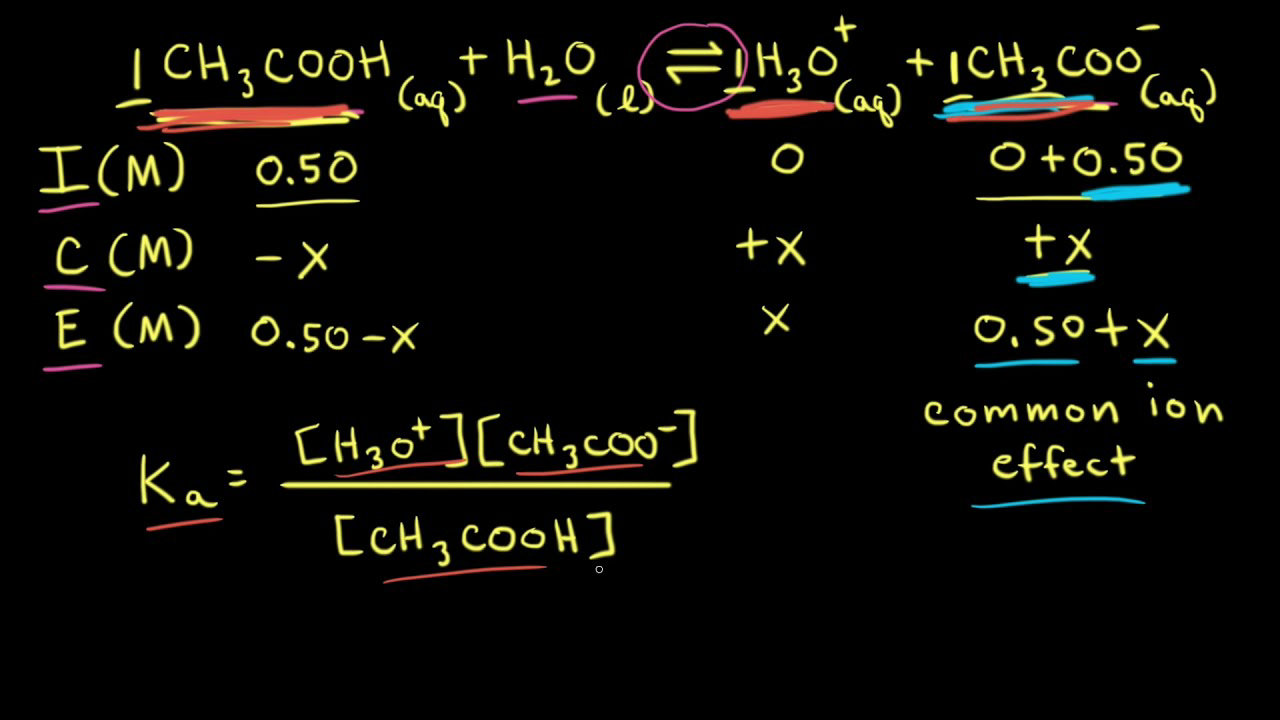
Worked example: Calculating the pH after a weak acid–strong base reaction (excess acid) (video) | Khan Academy